Flexible bronchoscopy (FB) is generally considered a safe procedure, even in critically ill, mecha-nically ventilated patients. However, FBs in the intensive care unit (ICU) carry several risks, such as accidental extubation, infection, progressive ate-lectasis, pneumothorax, bronchial injury, bleeding, and hemodynamic disturbances. Moreover, patients treated in the ICU may have a greater risk of complications due to underlying hypoxemia, hemodynamic instability, preexisting infections, and susceptibility to bleeding, as well as difficult technical and clinical conditions typical of the ICU environment [1–7]. As FBs are increasingly used in ICUs as diagnostic and therapeutic tools, there is a risk of overuse or misuse, leading to adverse events. In recent years, several excellent review articles have been published covering a wide range of issues related to FB in critically ill patients [8–14]. In this narrative review, we focused on the practical and safety aspects of performing FB in patients undergoing mechanical ventilation and on the technique of the most commonly performed interventions.
Selection of bronchoscopes and indications for bronchoscopy in the ICU setting
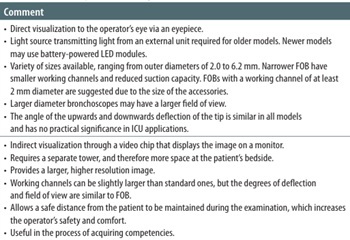
The choice of flexible bronchoscope in the ICU depends on local availability, the purpose of the procedure (therapeutic or diagnostic), ope-rator skill, the urgency of the procedure (i.e., life-threatening situations) and the space available at the patient’s bedside. The characteristics of the types of flexible bronchoscopes and selection guidelines are presented in Table 1. The most common indications for FB in the ICU are summarized in Table 2.
TABLE 1
Characteristics of the types of flexible bronchoscopes and selection guidelines
TABLE 2
The most common indications for flexible bronchoscopy in the ICU
Complications: fever and infection
Microorganisms originating from the upper respiratory tract may be transferred to the lower respiratory tract during oral bronchoscope insertion and may enter the bloodstream [17, 18]. Bacteremia after bronchoscopy is transient and occurs in approximately 5% of patients. Postbronchoscopy fever may occur in up to 50% of patients [19]. The release of cytokines from alveolar and inflammatory cells during bronchoscopy, which is caused by lavage, lidocaine instillation, and physical discomfort (e.g., challenging/traumatic manipulations, suction), is one potential cause of the development of fever. Although there are few clinical studies on this topic, the rate of postbronchoscopy pneumonia may approach 6% [20]. Overall, the risk of patient contamination is estimated at 15%, and the risk of infection following bronchoscopy is estimated at 2.8% [15]. According to certain clinical studies, broncho- fiberoscopy is a separate risk factor for ventilator-associated pneumonia (VAP) [21, 22]. The introduction of biofilm-enclosed bacteria into the lower respiratory tract in the context of the endotracheal tube (ETT), which inhibits mucociliary clearance, is one potential mechanism by which bronchoscopy may predispose patients on mechanical ventilation to VAP. Given that colonization and biofilm formation on the ETT usually start a few hours after intubation and that it takes three to seven days for bacteria to be isolated from tracheal aspirates, such a mechanism is theoretically possible [23, 24]. Modern FBs contain multiple channels and ports that can easily collect organic material [25]. Even if proper endoscope reprocessing protocols are followed, the accumulation of microorganisms can, over time, lead to the development of biofilms within the narrow channels of the endoscope [25–28]. Inaccessible biofilm formation on the internal surface of endoscope channels, especially when they are scratched or damaged, can result in failure of the reprocessing process. This can create a vicious cycle of contamination, growth, disinfection, and regrowth, increasing the risk of bacterial transmission [18]. Several outbreaks and pseudo-outbreaks of multidrug- resistant organisms (MDROs) that are strongly linked to the contamination of bronchoscopes have been reported [29–31], indicating that bronchoscopes may have an underestimated potential for MDRO transmission, justifying increased awareness among healthcare workers, improved preventive measures, and strict adherence to reprocessing recommendations [25].
Most bronchoscopes are classified as semicritical devices requiring at least high-level disinfection (HLD), which implies the complete elimination of all microorganisms in or on an instrument, except for a small number of spores. Although guidelines recommend sterilization of semicritical devices when feasible, HLD of bronchoscopes has been routinely practiced for decades and remains commonplace, primarily because endoscopes may be damaged by heat [25, 32, 33]. The optimal frequency of culturing a bronchoscope for contamination with MDROs is unknown. However, routine pathogen isolate review to identify unexpected clusters or trends is recommended [25, 28, 30]. When bronchoscopes are kept in transport cases or unventilated cabinets, there is a chance that moisture will build up and cause bacterial and fungal growth. Flexible bronchoscopes should be stored in a spacious cabinet with sufficient ventilation and hung upright without valves when not in use. Only when bronchoscopes are transported over long distances should carrying cases be considered [31, 34, 35]. When institutional standards for reprocessing cannot be completely met, single-use bronchoscopes are advised.
Studies have shown that bronchoscopy is an aerosol-generating procedure that exposes medical personnel to tiny and respirable aerosols for brief periods of time, increasing the risk of infection transmission. Guidelines for the use of personal protective equipment (PPE) have been updated as a result of the increased concern of the COVID-19 pandemic [36]. Current PPE protocols should be adapted to the current local COVID-19 prevalence and institutional infection control policies. Although universal precautions (gloves, gowns, and eye protection) remain standard for all bronchoscopies, N95 respirators or equivalent respirators should be used on the basis of current risk assessments [36].
Procedural sedation and topical anesthesia
The vast majority of critically ill patients who undergo FB in the ICU are already sedated according to protocols adopted in individual centers. However, it may be necessary to deepen sedation to control coughing, body movements, and autonomic reactions. Sedation should be tailored to patient comfort and safety, with local anesthesia as the primary method of cough suppression. Deep sedation may be reserved only for select patients who remain intolerant despite adequate topical anesthesia with careful monitoring of hemodynamic and respiratory status [37]. Neuromuscular blocking agents may play a role during FB to facilitate its performance, primarily in situations where deep sedation is desired in hemodynamically unstable patients. However, few clinical studies have focused on this topic. The deepening of sedation in critically ill patients is usually achieved by administering additional doses of continuous infusion drugs (e.g.,propofol, midazolam, dexmedetomidine, ketamine). The addition of an opioid to midazolam or propofol improves procedural cough, reduces lidocaine usage, and increases patient procedural tolerance [38].
Effective topical anesthesia blunts gags, cough, and laryngospasm. A reduction in cough improves patient comfort and makes bronchoscopy safer for the patient and easier for the operator. The underlying mechanisms of cough suppression with local anesthetics are not clearly understood [39]. Sedation should be titrated to increase patient comfort, but in some high-risk patients, local anesthesia alone may be safer [40]. In an intubated patient, lidocaine may be administered to the lower airways via ‘spray-as-you-go’ delivery of a 1–2% solution via the bronchoscope working channel directly into the airway [41]. One percent lidocaine was found to be as effective as a 2% solution for as-you-go topical anesthesia during FB, albeit at a significantly lower dose as the latter. Thus, 1% may be the preferred concentration for topical anesthesia during FB. Lidocaine may also be administered intravenously within a single bolus range of 0.5–2 mg kg−1 to inhibit the cardiovascular response, cough reflex, and cardiac arrhythmias and increase intracranial and intraocular pressure. Intravenous administration of lidocaine may be indicated in patients with heart failure, conditions causing increased intracranial pressure (including traumatic brain injury), and obstructive pulmonary disease [39, 42, 43]. The different routes of administration are not mutually exclusive, but care should be taken not to exceed the maximum cumulative dose. Special caution should be exercised in patients with renal impairment, hepatic dysfunction, and congestive heart failure due to the ease of lidocaine overdose.
Lubricants
The most frequent damage to flexible optic bronchoscopes (FOBs) during procedures in intubated patients involves the rubber coat of the distal bending section. They usually result from wear and tear of the external sheath due to rough manipulation [44]. Lubricants are a necessary adjunct to facilitate the passage of the bronchoscope through the rigid edges and surfaces of tubing, connectors, and other components in contact with the bronchoscope [36]. Water-soluble lubricants are most often recommended, whereas petroleum-based products should be avoided. The latter may cause premature wear, stretching, and deterioration of the rubber sheath of the bronchofiberscope [45]. Moreover, repeated exposure of mucous membranes to hydrocarbon compounds of high viscosity and surface tension, such as mineral oils and paraffin, may cause chronic and acute forms of lipoid pneumonia, including adult respiratory distress syndrome [45]. Various other lubricants have been used in some facilities, e.g., simethicone, vegetable oils, cooking oil sprays, intimate hygiene gels, and tissue glue, which are not labelled for medical use. Leading manufacturers warn customers against the use of petroleum-based lubricants (e.g., paraffin, petroleum jelly) and silicone [45]. Although these lubricants may not damage the endoscope coat, they can be difficult to remove from the channels during reprocessing and provide a safe haven for biofilms [28, 46]. Lidocaine gel is likely the most commonly used lubricant in the critical care setting. Products available on the market often contain, in addition to local anesthetics, propylene glycol and hydroxyethylcellulose as lubricants and chlorhexidine gluconate as an antiseptic. Their advantages include sterility, ease of application, and effective reduction of friction. However, they can dry out during longer procedures, which necessitates reapplication. Theoretically, lidocaine overdose can also occur if topical anesthesia is used in parallel. Alternatively, silicone spray labelled for medical use is commonly used in some facilities. When applied carefully, it is effective in reducing friction and preventing FB damage, and appears to be microbiologically safe.
Endotracheal tubes
FB has a slight to moderate impact on patients’ respiratory flow, work of breathing, and airway resistance in nonintubated patients. However, individuals on mechanical ventilation experience notable alterations in respiratory mechanics. Depending on the ventilation mode and the previously set inspiratory pressure limit, minute ventilation drops, and positive end-expiratory pressure increases as soon as the bronchoscope is inserted into the ETT [7, 47, 48]. The peak inspiratory pressure can reach up to 80 cmH2O, but this represents ventilator backpressure rather than true intratracheal pressure. The peak tracheal pressure ranges from 18 to 60 cmH2O, which is closely related to the internal diameter of the ETT [7]. The intrinsic PEEP, which is caused by decreased expiratory flow and insufficient lung emptying during expiration, is represented by the latter pressure. Owing to reduced venous return, barotrauma, and possibly pneumothorax, FB-induced hyperinflation may cause hemodynamic compromise. Additionally, because of the loss of lavage fluid and bronchospasm caused by tracheal stimu-lation, FBs may generate zones of poor ventilation–perfusion ratios and a minor intrapulmonary shunt [49]. High airway pressures are closely associated with the ETT diameter, specifically the diffe-rence between the internal diameter of the ETT and the external diameter of the bronchoscope, according to experimental research. Approximately 40% of the 9 mm diameter ETTs and two-thirds of the 7 mm ETTs make up a bronchoscope with a diameter of 5–6 mm. The intrinsic PEEP can exceed 35 cmH2O during bronchoscopy with a smaller diameter tube, greatly increasing the risk of complications [7, 50]. Additionally, patients who are “fighting” with ventilators have a significantly greater risk of barotrauma [7].
It is generally recommended to maintain a difference of at least 1.5-2 mm between the internal diameter of the ETT and the diameter of the flexible bronchoscope or to use at least 8 mm of tubing to prevent high airway resistance [9, 14, 51]. In clinical practice, attention should be given to narrowing the ETT because dried secretions or blood clots impair its patency. Replacement of the tube before the procedure may be indicated in such situations. Another way to avoid hyperinflation may be to use FBs with a smaller external diameter. In this case, however, interventions may be more difficult due to the visualization conditions and the narrower working channel of the device [48]. FB may be technically easier with a tracheostomy tube than with an ETT because of the shorter tube length and lower insertion resistance. However, a greater bending angle can be problematic, especially with smaller tracheostomy tubes. Maintaining an appropriate distance between the inner tube diameter and the FB diame-ter also applies to tracheostomy tubes.
Mode of ventilation
The primary concern when performing FB in patients in the ICU is hypoxia and, to a lesser extent, hypercarbia [2, 9, 37]. The risk of hypoxemia is related to the patient’s baseline oxygen saturation (SaO2), lung function, and comorbidities. In addition, the type and mode of procedure, duration of the procedure, mode of ventilation used, volume of lavage and depth of sedation may affect gas exchange [2, 9, 37, 48].
There are currently no evidence-based recommendations for the selection of the ventilation mode during FB in critically ill patients. Expert opinion suggests that volume-based (VC) modes may be preferred [9, 14]. Nevertheless, airway resistance increases when the bronchofiberscope is introduced into the ETT. If the peak pressure limit is not raised, the ventilator will not deliver the predetermined tidal volume, resulting in hypoventilation [51]. However, there is a risk that the intrinsic PEEP will rise uncontrollably regardless of the ventilation mode selected. Expiratory flow is impeded by a bronchofibroscope that takes up a significant portion of the ETT lumen, and an air trap may develop with an uncontrollably high intrapulmonary pressure, particularly at higher ventilatory rates [51]. Lowering the initial PEEP level could be a potential strategy for reducing the risk of barotrauma [14]. Nevertheless, blind PEEP correction could be hazardous since there are currently no instruments available to detect airway pressure directly in ventilator-independent circumstances [48]. Leakage of the connector’s sealing ring and suction via the working channel may partially mitigate the intrinsic PEEP effect [7]. In clinical practice, it would seem reasonable to maximize the exhaled tidal volume with the bronchofiberscope in place before beginning the real procedure by adjusting the peak pressure value, respiratory rate, and inspiratory-to-expiratory (I:E) ratio [9, 48]. Additionally, the low-minute volume alarm level needs to be checked and adjusted. It is advisable to stop the procedure and use recruitment techniques to help the patient return to their normal ventilation and gas exchange parameters if hypoventilation continues despite all the interventions. Crucially, bronchoscopy in critical care patients is a dynamic procedure in real life. In critically ill patients, bronchoscopy procedures should be carried out in short sequential cycles with continuous suction intervals limited to a few seconds to lower the danger of derecruitment or increase the intrinsic PEEP [52, 53]. Patients with restrictive disorders (e.g., acute respiratory distress syndrome) require precise lung-protective-ventilation (LPV) strategies. In this group of patients, bronchoscopy can be particularly challenging, resulting in significant variations in airway pressures and leading to disruption of LPV [48, 54]. Considering the increase in respiratory resistance after the bronchoscope is inserted, it is reasonable to titrate the inspiratory pressure to a level that allows an appropriate tidal volume to be obtained. If the PC mode is used during the procedure, it is critical that the inspiratory pressure level be lowered to baseline, i.e., preprocedural values, before the bronchoscope is removed from the ETT. Failure to do so exposes the lungs to high inspiratory pressures, hyperinflation, and barotrauma [51].
Although prone positioning is a primarily lung-protective maneuver, it may result in the mobilization of secretions into the airways, impairing oxygenation and providing a nidus for infection [55]. Despite several risks, FB may be beneficial in this situation. To date, few case reports demonstrate that prone bronchoscopy is feasible and effective in achieving the clinical goal, provided that enhanced safety criteria are applied to reduce the risk of derecruitment or increase end-expiratory pressure [6].
Owing to concerns about the possible risk of transmission of the SARS-CoV-2 virus to medical personnel, the apnea-intermittent bronchoscopy technique was proposed [56]. Patients are deeply sedated, and after careful preoxygenation, the respiratory system is disconnected. The bronchoscope was inserted directly through the ETT, and the procedure was continued under apnea. If the oxygen saturation decreases below 90%, bronchoscopy is interrupted, and the patient is reconnected to the ventilator. After an additional period of preoxygenation, bronchoscopy is then completed. Although this protocol differs from common practice, the procedure has been shown to be effective, safe and well tolerated by patients. In our center, bronchoscopy with the intermittent apnea technique is used in situations where it is necessary to avoid uncontrolled increases in airway pressure, e.g., in patients after lung resection procedures. However, this technique has never been validated and cannot be recommended for routine use.
Lavage and suction
Although diagnostic bronchoalveolar lavage (BAL) is considered safe and well tolerated by ventilated, critically ill patients, some patients may experience prolonged hypoxemia following this procedure [57, 58]. During BAL, abundant aspiration leads to a significant, although transitory, decrease in lung volume and lung compliance with an increase in respiratory resistance [4, 59]. A decrease in the PaO2/FiO2 ratio > 25% may occur in up to 30% of ICU patients, with a reduction in blood oxygen saturation (SaO2 ≤ 88%) and/or bronchospasm in 10% of patients 1 h after BAL. Hemodynamic instability in the first 24 h after BAL may occur in approximately 20% of patients, especially in the case of concomitant cardiovascular diseases [2, 58–61]. Therefore, the potential benefits of BAL should be weighed against the invasiveness of the procedure and the risk of severe complications. FB and BAL procedures should not be performed in patients with a deplorable clinical condition [2]. Unfortunately, no clear-cut clinical criteria for withholding FBs or BALs have been defined to date. A structured approach that takes into account additional medical staff is necessary to ensure a high level of procedural safety.
Available methods to minimize contamination of BAL fluid with upper respiratory tract secretions should be used. Suctioning or injecting lidocaine through the working channel of the bronchoscope during insertion should be avoided. Suctioning through the ETT should be performed prior to bronchoscopy. The first 20 mL of sterile saline should be instilled to clear any residual upper airway secretions and aspirated separately (should not be subjected to quantitative bacterial culture or microscopic evaluation). The bronchoscope tip should be positioned just above the lung segment or subsegment from which the sample is to be collected. A collection device is connected to the bronchofiberscope, and additional aliquots of 20 to 60 mL are instilled and aspirated back after each instillation. The total amount of fluid injected should be 120–140 mL [2, 9, 62]. The amount of fluid required to obtain an adequate sample and the amount of recovered fluid required for adequate microbiological analysis are not consistent. One study reported that a > 40% increase in the lavage volume was associated with increased VAP [62]. Often, additional suctioning may be necessary to achieve an adequate return volume. Other maneuvers, such as pulsatile suctioning, patient repositioning, ventilator adjustments, and bronchoscopic maneuvers, may aid collection. Using additional fluid outside of the protocol is unlikely to improve the return rate and carries the risk of diluting colony counts [62].
Clot removal
Acute airway obstruction, due to blood clots, mucus plugs, or foreign bodies, is a life-threatening emergency in the ICU. Airway obstruction can result from several conditions, including inhalation burns, infectious tracheobronchitis, suctioning trauma, vascular anomalies, and coagulation disorders (including ECMO-related disorders), as well as after tracheostomy, airway stenting, and cardiothoracic surgery [63–65]. Initial measures to remove endobronchial plugs include lavage and suction, with the removal of small plugs adhering to the tip by continuous suction. In the case of larger conglomerates, defragmentation may be necessary, usually with biopsy forceps, basket extraction, or balloon catheter dislodgement. Local application of thrombolytic agents followed by suction may be helpful if clots are firmly attached to the mucosa [65–68]. Thrombolytic agents may, however, pose a risk of rebleeding, whereas manipulation with forceps or a basket may cause additional damage to the bronchial mucosa. Manipulation of the Fogarty catheter around the clot is a blind procedure that itself may cause injury. In addition, these techniques are time-consuming and not always feasible in patients with severe respiratory failure.
Recently, flexible bronchoscopic cryoextraction, which is performed bedside in the ICU, has become a valuable alternative for the successful removal of blood clots and mucus plugs. Direct contact with the cryoprobe tip freezes the liquid component of blood clots, airway secretions, and foreign bodies. A liquid cryogen or coolant (nitrous oxide or carbon dioxide) is delivered under pressure to a specially designed cryoprobe that can be passed through the working channel of the bronchoscope. The cryoprobe is applied in direct contact with the clot or plug, and freezing is performed until the point of contact is frozen, causing adherence to the probe, usually for 10 seconds. Care must be taken to avoid contact of the probe with the airway mucosa while cooling. In the next stage, both the bronchoscope and the probe are removed together, with the adhered clot attached to the tip. Successful cryoextraction has been reported in several case reports and retrospective studies, with a success rate of 92–100%. The entire procedure usually takes less than ten minutes to complete [63, 64, 67]. The use of a cryoprobe may have a be-neficial hemostatic effect by constricting blood vessels and reducing bleeding, but the evidence is limited [64, 67]. Notably, fluid may freeze in the working channel in the narrow space surrounding the cryoprobe and rupture the inner lining of the channel. To avoid this problem, the cryoprobe tip should be at least 4 to 5 mm distal to the bronchoscope tip when freezing. Complete thawing (e.g., in warm water) should occur before withdrawing the cryoprobe through the channel.
Regardless of the tactics adopted, every effort should be made to avoid pushing the foreign body distally into the airway during the removal procedure. The key to removing clots is the ability to adequately secure the object by grasping it or enclosing it with forceps or a basket. In the case of mobile plugs, an attempt may be made (e.g., using a Fogarty balloon) to pull the object in a retrograde manner, bringing it proximally into the trachea before attempting removal. Once the object is grasped, all three (bronchoscope, grasping instrument, and clot) are removed from the patient simultaneously as a unit. During removal, every effort should be made to maintain visual contact with the clot, keeping it in the center of the airway. In the case of large clots that do not fit through the ETT, en bloc removal of the clot with the ETT may be necessary [65, 67, 68]. In such cases, one should be prepared for rapid reintubation, ideally assisted by an experienced anesthesiologist, who, during removal, exposes the larynx with a laryngoscope and immediately inserts a new ETT.
Special situations
In patients with out-of-hospital cardiac arrest (OHCA) and return of spontaneous circulation (ROSC), loss of natural airway clearance, possible aspiration, lung contusion, coma, therapeutic hypothermia, and mechanical ventilation are risk factors for pneumonia. These patients might have better outcomes if they receive bronchoscopy early after hospital admission. The reported data suggest that early bronchoscopy is associated with a shorter intubation period [69].
The diagnosis of inhalation burns is based on a history of exposure to fire and smoke in a confined space associated with impaired consciousness. Bronchoscopy performed within the first 24 hours confirms the diagnosis, assesses the severity of the injury, and allows for a good prognosis [70, 71]. However, serial bronchoscopies performed for assessment of the progression of inhalation injury may provide more risk than benefit [71, 72]. Serial surveillance of the airways and therapeutic lavage may be considered in severe burn inhalation injury [72].
The main use of FBs in patients with aspiration pneumonitis is removal of aspirated material and sampling of the lower respiratory tract. Early bronchoscopy (within 24 h of intubation) has been demonstrated to improve respiratory function, decrease the incidence of aspiration pneumonia, and guide the de-escalation of antibiotic therapy [73, 74].
Summary
FB is a safe procedure for critically ill patients only if the operator understands and adheres to safety considerations. The decision to perform FB in high-risk patients requires a careful risk-benefit balance. The operator must be aware that clinical deterioration may occur during the procedure; thus, the procedure should be performed in a properly equipped environment. Internal procedures, including minimum operator qualifications, patient and equipment preparation, examination techniques, and troubleshooting, should be implemented and followed. A comprehensive, FB ICU checklist is highly recommended (Figure 1).