The concept of de-resuscitation has gained increasing attention in critical care medicine, representing the active removal of excess fluid once the acute resuscitation phase has been completed [1, 2]. The identification of patients who would benefit from de-resuscitation requires accurate assessment of fluid overload and the potential for safe fluid removal, a process that relies heavily on the integration of clinical assessment, objective measurements, and careful monitoring of response to interventions [3, 4]. Historically, fluid assessment in the intensive care unit (ICU) has relied heavily on clinical examination findings, including the presence of peripheral oedema, pulmonary crackles, elevated jugular venous pressure, and weight gain [5–7].
However, randomised controlled trials and systematic reviews investigating de-resuscitation have failed to demonstrate consistent patient benefit [8–10]. A possible reason could be the fact that such trials use parameters such as cumulative fluid balance and the presence of oedema as triggers to commence de-resuscitation. The relationship between clinical assessment findings and objective measures of fluid status has been the subject of considerable research, but the results have been notably inconsistent and often contradictory [11–14].
Point-of-care ultrasound (POCUS) has revolutionized many aspects of critical care medicine, offering real-time, non-invasive assessment of cardiovascular function, lung pathology, and fluid status [15, 16]. Through US assessment of biventricular function, lung and pleural spaces, as well as venous compartments, it is now possible for clinicians to build a more complete picture of fluid status [17, 18].
The objective of this study was to determine, in critically ill patients who had clinical signs of fluid overload, the incidence of US assessed markers of fluid overload and their correlation with traditional clinical findings.
Methods
Study design and setting
The INCIdence of abnormal VEnous ultrasound in patients with eXcess fluid (INCIVEX) study was an observational, cross-sectional study conducted at a tertiary-level mixed ICU.
Ethical considerations
The study was conducted in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice guidelines. The study met the criteria for a service evaluation according to the UK NHS Health Research Authority tool, and national ethical approval was not required; approval was provided by the local hospital committee (King’s College Hospital, Department of Critical Care). Given its observational nature and use of standard clinical assessment techniques, the research was classified as minimal risk, and written informed consent was waived.
Study population and inclusion criteria
Inclusion criteria were adult patients (> 18 years), admitted to the ICU for at least 72 hours, exhibiting clinical evidence of fluid overload but no evidence of circulatory malperfusion. Fluid overload was defined by the presence of oedema and a cumulative positive fluid balance exceeding 2 L [19, 20]. Additionally, patients were required to be haemodynamically stable, defined as a systolic blood pressure > 90 mmHg, with minimal vasopressor support (noradrenaline equivalent dose ≤ 0.2 µg kg–1 min–1), and a lactate level below 2.0 mmol L–1.
Patients were excluded from the study if they were undergoing active resuscitation or experiencing haemodynamic instability, had an acute myocardial infarction within the previous 48 hours, or had known severe valvular heart disease that would interfere with cardiac US assessment. Additional exclusion criteria included chronic kidney disease requiring renal replacement therapy prior to ICU admission, known chronic heart failure with an ejection fraction less than 30%, pregnancy, or designation for comfort or palliative care.
Data collection
Baseline participant demographics were collected at study inclusion. Clinical and physiological parameters were recorded, including mechanical ventilation status, presence and location of oedema, cumulative fluid balance from ICU admission, and vasoactive medication type and dose (noradrenaline dose and number of vasopressors).
US assessment
US assessment was performed using standardized protocols by physicians trained in POCUS techniques, achieving the equivalent of the UK FUSIC accreditation level. All assessments were performed using a GE Venue (Boston, USA) or Mindray TEX20 (Shenzhen, China). All images and video sequences were de-identified and reported by an assessor blinded to the clinical situation.
Focused cardiac US: Standard 4-chamber views were digitally recorded for offline analysis. Para-meters measured included tricuspid annular plane systolic excursion (TAPSE), mitral annular plane systolic excursion (MAPSE), left ventricular (LV; LVOT VTi, S’) and right ventricular (RV) function (assessed qualitatively as ‘normal’ or ‘abnormal’), and peak tricuspid regurgitation velocity. Normal TAPSE and MAPSE were defined as more than 16 mm and 10 mm, respectively.
Lung US: Lung US was performed according to established protocols, dividing the chest wall into six zones. The presence or absence of B-lines, pleural sliding, lung consolidation, and pleural effusions was recorded for each zone [21–23].
Abdominal US and Venous Excess Ultrasound (VExUS) grading system: Assessment included the inferior vena cava (IVC), hepatic vein, portal vein, and intrarenal vein using 2D as well as Doppler analysis. VExUS scores were calculated as described else-- where [24].
Statistical analysis
Statistical analysis was conducted using Prism v 10.4 (GraphPad Software, San Diego, CA).
Given the exploratory nature of this study, we adopted a convenience target recruitment of 50 pa- tients.
Descriptive statistics were calculated for all variables, with continuous variables presented as means ± standard deviations or medians with interquartile ranges depending on distribution charac-teristics. Normality of continuous variables was assessed using the Shapiro-Wilk test. Categorical variables were presented as frequencies and percentages.
For associations between continuous variables, Spearman’s rank correlation coefficients were calculated with 95% confidence intervals. For associations between categorical variables, Fisher’s exact test was used due to the relatively small sample size. For associations between continuous and cate-gorical variables, the Mann-Whitney U test was employed. Statistical significance was defined as a two-sided P-value < 0.05.
Results
Study population characteristics
In total, 195 patients were screened during a 2-month period. Of these, 54 patients met the inclusion criteria and were recruited to the study. The US images of 4 of these patients were suboptimal and precluded analysis; these patients were excluded from the final analysis.
The mean age of patients was 58.6 ± 15.9 years, with a predominance of males (70.0%, n = 35). The demographic and clinical characteristics of the study population at baseline are presented in Table 1.
TABLE 1
Patient demographics and baseline characteristics
The study population exhibited significant diversity in terms of primary admission diagnoses, with sepsis and septic shock representing the most common indication for ICU admission (36.0%, n = 18), followed by respiratory failure (22.0%, n = 11), cardiovascular emergencies (18.0%, n = 9), and post-operative complications (16.0%, n = 8).
Fluid balance and clinical parameters
Cumulative fluid balance calculations revealed substantial fluid accumulation in the study population, with a mean cumulative positive balance of 7,200 mL (SD ± 4,800) at the time of inclusion.
Assessment of clinical fluid overload indicators revealed a high prevalence of peripheral oedema in the study population. Upper limb oedema was pre-sent in 66.0% of patients (n = 33), while lower limb oedema was observed in 74.0% of patients (n = 37).
US assessment findings (Table 2)
LV dysfunction was identified in 16.0% of patients (n = 8), with the majority exhibiting mild to moderate impairment. RV dysfunction was observed in 10.0% of patients (n = 5), with findings including RV dilatation, reduced systolic function and abnormal interventricular septal motion.
TABLE 2
Ultrasound findings
Lung US assessment revealed evidence of pulmonary fluid accumulation in nearly half of the patients. Bilateral pleural effusions were present in 44.0% of patients (n = 22), with effusion volumes ranging from small to moderate in most cases. The presence of B-profile, defined as ≥ 3 B-lines in at least two zones, was observed in 12.0% of patients (n = 6).
The VExUS protocol assessment provided systematic evaluation of venous congestion across multiple vascular territories. IVC assessment revealed dilated IVC (> 2 cm diameter) in 36.0% of patients (n = 18). Hepatic vein profiles showed the lowest degree of congestion, with 41 patients (82.0%) demonstrating Grade 0, 6 patients (12.0%) Grade 1 and 3 patients (6.0%) Grade 2 congestion. Portal vein profiles exhibited more heterogeneous distribution: 25 patients (50.0%) had Grade 0, 17 patients (34.0%) Grade 1 and 8 patients (16.0%) Grade 2 congestion. Renal vein profiles predominantly showed minimal congestion, with 38 patients (76.0%) having Grade 0, 11 patients (22.0%) Grade 1, and only 1 patient (2.0%) Grade 2 congestion. The overall VExUS score distribution showed that 20.0% of patients (n = 10) had elevated scores (≥ 2, moderate-severe congestion).
Correlation analysis
Relationship between fluid balance, oedema, and US parameters
Cumulative fluid balance showed no significant associations with any US or echocardiographic parameters (n = 50, all P > 0.05).
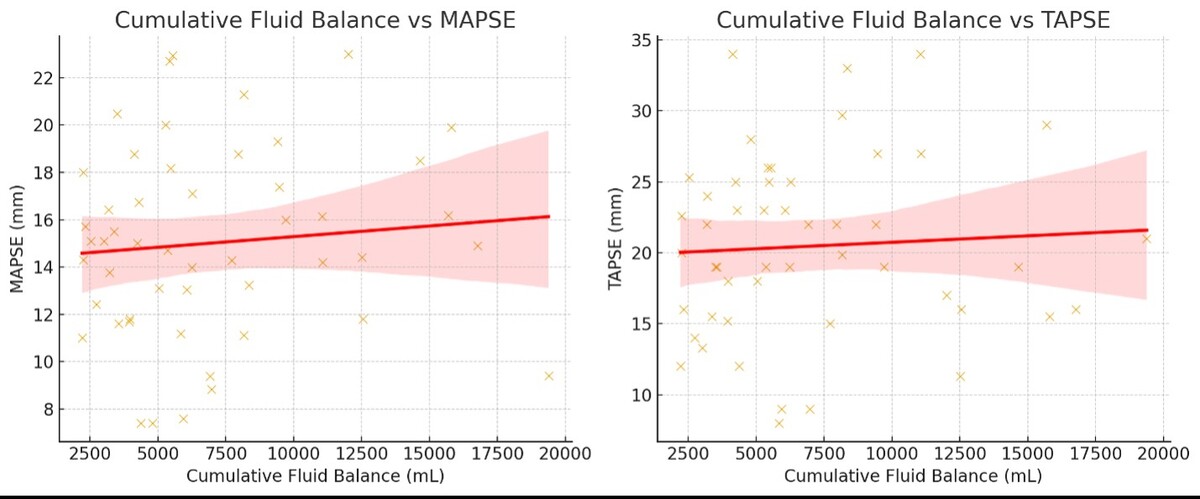
No significant correlations were observed between cumulative fluid balance and qualitative assessment of either LV or RV function. There was no significant association between cumulative fluid balance and either MAPSE (ρ= 0.11, confidence interval –0.188 to 0.364, P = 0.45) or TAPSE (ρ= 0.10, confidence interval –0.221 to 0.334, P = 0.47) (Figure 1).
FIGURE 1
Relationship between cumulative fluid balance and (A) mitral annular plane systolic excursion (MAPSE)/(B) tricuspid annular plane systolic excursion (TAPSE)
The group of patients with LV dysfunction had similar fluid balance (median 6593 mL, IQR 1880) compared to the group without (median 5451 mL, IQR 5880), P = 0.631. The group of patients with RV dysfunction had similar fluid balance (median 5928 mL, IQR 3877) compared to the group without (median 5546 mL, IQR 5281), P = 0.593.
No significant correlations were observed between cumulative fluid balance and venous congestion scores. Spearman correlation coefficients were: hepatic vein profile (ρ= 0.071, P = 0.626), portal vein profile (ρ= –0.144, P = 0.319), renal vein profile (ρ= –0.114, P = 0.431), and VExUS (ρ= –0.162, P = 0.264).
Associations between cardiac and lung US with venous congestion
RV dysfunction emerged as the strongest predictor of systemic venous congestion, with affected patients demonstrating significantly higher hepatic vein congestion scores (median 1.0 vs. 0.0, P = 0.003), renal vein congestion scores (median 1.0 vs. 0.0, P = 0.033), and VExUS scores (median 2.0, P < 0.022).
In contrast, LV dysfunction showed mixed associations, with paradoxically lower portal vein congestion scores (median 0.0 vs. 1.0, P = 0.023) but higher renal vein scores (median 0.5 vs. 0.0, P = 0.048).
Lung US findings, including bilateral pleural ef-fusions and B-profile patterns, demonstrated no significant associations with any venous congestion parameters (all P > 0.05), indicating that pulmonary and venous US assessments provide complementary rather than duplicate information in the evaluation of fluid overload.
Discussion
The INCIVEX study highlights a key observation: cumulative fluid balance, despite large positive values, is not correlated with either clinical signs of fluid overload or US-based measures of cardiac dysfunction, pulmonary congestion, or venous congestion. This potentially provides a plausible explanation as to why prior de-resuscitation trials, designed around fluid balance or clinical thresholds, have not consistently shown benefit. The fundamental disconnect challenges the validity of long-standing tools for fluid status assessment in the ICU, with important implications for both clinical practice and research trial design.
These findings may explain why multiple randomised controlled trials of de-resuscitation have failed to demonstrate consistent benefit. Landmark studies have often relied on cumulative fluid balance thresholds or the presence of oedema as entry criteria or therapeutic targets [1, 4, 19, 25–27]. Our study shows that such markers are dissociated from US-based fluid status. This raises the possibility that a considerable proportion of patients enrolled in prior trials may not have had true fluid intolerance, a relatively new concept [28, 29], while others with significant congestion were overlooked. This may have diluted any potential treatment effect.
The limitations of cumulative fluid balance are both methodological and physiological. From a methodological standpoint, charts are prone to error and incomplete recording, and fail to capture insensible or “hidden” losses. Physiologically, fluid balance calculations cannot reflect the complex distribution of fluid in critical illness, where capillary leak, glycocalyx disruption, and altered lymphatic drainage uncouple administered volume from clinically perceived fluid accumulation [30–32]. For example, evaluation of fluid status based on the presence of oedema alone is problematic, as peripheral oedema reflects interstitial fluid accumulation at tissue level. This clinical sign provides little insight into central venous pressures, organ congestion, or the ability of the cardiovascular system to tolerate the addition or removal of fluid.
POCUS offers a real-time assessment of cardiovascular function and fluid tolerance that directly addresses the limitations of traditional methods [33–36]. Cardiac US provides immediate evaluation of ventricular function and filling status, while lung US can detect pulmonary oedema with superior sensitivity to clinical examination or chest radio-graphy. The INCIVEX study demonstrates that US and clinical findings should be viewed as complementary, each capturing different facets of fluid physiology; it underscores the importance of a multimodal approach.
We propose that clinical practice should evolve toward integrated assessment strategies that combine clinical context with objective haemodynamic information over traditional fluid balance calculations, which will potentially improve patient selection and therapeutic outcomes [37]. Development of US-guided de-resuscitation protocols, incorporating cardiac function assessment, lung US, and venous congestion evaluation, would represent a paradigm shift from volume-based to physiology-based fluid management [12, 38].
The implications for future research are clear. It should be noted that trials investigating the use of POCUS and fluid management have tended to examine fluid responsiveness and resuscitation rather than de-resuscitation [39, 40]. De-resuscitation trials must move beyond reliance on crude measures such as cumulative fluid balance to incorporate objective physiological inclusion criteria and study endpoints. Patient selection based on US evidence of fluid intolerance, such as RV dysfunction, IVC dilatation, or venous congestion, may enrich study populations with individuals perhaps more likely to benefit from fluid removal. Likewise, trial success should be measured not by numerical changes in fluid balance, but by improvements in organ function, haemodynamics, and patient-centred outcomes.
The VExUS protocol, despite limited correlation with clinical signs in our study, may provide valuable information about venous congestion and organ perfusion that is not captured by traditional assessment methods [41–43]. Other studies have shown that VExUS can be used to track the de-resuscitation process [44, 45]. In the clinical setting, it is likely that the utility of VExUS may be as part of an integrative, multi-modal assessment of both fluid responsiveness and congestion [46, 47].
Our study has limitations. It was a single-centre study of a modest sample size, limiting the precision of correlation estimates and generalisability. The cross-sectional design precludes insight into the temporal dynamics of fluid distribution. Nevertheless, by systematically comparing clinical, fluid balance and US assessments in a well-defined population, our study provides important evidence that traditional metrics are inadequate guides for de-resuscitation. In addition, we did not measure the patients’ body weight, as it is not usual practice in our ICU. The changes in body weight is another established parameter to track fluid status, with its own set of limitations [48].
Conclusions
The INCIVEX study demonstrates that cumulative fluid balance and clinical signs of oedema do not correlate with objective US markers of fluid status in critically ill patients. POCUS offers physiologically meaningful insights, although its relationship to traditional assessment must first be clarified before it can be fully integrated into practice.
De-resuscitation strategies should move toward physiology-based methods, using clinical context and objective assessments to guide fluid removal for appropriate patients and improve outcomes.