Percutaneous dilatational tracheostomy (PDT) is essential in the intensive care unit (ICU) for patients struggling to wean from mechanical ventilation due to severe illnesses such as brain trauma or cerebrovascular events [1, 2]. The bedside feasibility of PDT, together with its favorable safety profile compared to open surgical tracheostomy, has contributed to its widespread adoption in the ICU setting [3, 4].
The optimal timing of PDT and its potential impact on clinical outcomes, including mortality, ICU length of stay (LOS), duration of sedation, and ventilator-associated pneumonia (VAP) incidence, have been a subject of ongoing debate in the literature. However, recent meta-analyses have yielded conflicting results [5–7].
Although PDT is less invasive than surgical tracheostomy [8], it may still trigger systemic inflammation in critically ill patients, potentially affecting outcomes. Notably, the role of the inflammatory response following PDT has not been extensively investigated in the literature, and it cannot be excluded that such a response might represent a factor influencing clinical outcomes, beyond the timing of the procedure.
Currently, the literature reports several indices, such as the neutrophil-to-lymphocyte ratio (NLR), the platelet-to-lymphocyte ratio (PLR), and the Systemic Inflammation Response Index (SIRI), that are capable of objectively assessing the inflammatory response triggered by a stressful event [9–14]. These hematologic indices represent the integrated output of the complex interactions between systemic stress and the effectiveness of the body’s homeostatic mechanisms. By summarizing different components of the innate and adaptive immune response, they offer a comprehensive view of inflammatory balance. Neutrophil-based ratios reflect the magnitude of the acute inflammatory reaction, whereas lymphocyteand monocyte-related components provide insight into immune competence and immune reserve. Their major advantage lies in the fact that they are easily derived from routine laboratory tests, without additional costs, making them readily applicable in daily clinical practice. When monitored over time, these indices may help detect inadequate or dys-regulated inflammatory responses, thereby identifying patients undergoing PDT who are at higher risk of complications or adverse outcomes. To date, no studies have specifically explored the temporal evolution of these indices in patients undergoing PDT.
This retrospective study aimed to assess changes in inflammatory parameters in ICU patients undergoing PDT. We investigated whether baseline and post-procedural values of these biomarkers are associated with mortality and whether their trends are prognostic for this high-risk group.
METHODS
We retrospectively reviewed medical records of ICU patients who underwent PDT between January 1, 2020, and June 10, 2024. All ICU admissions were recorded using the ProSafe system, a project supported by the Italian Group for the Evaluation of Interventions in Intensive Care Medicine (GiViTI, https://giviti.marionegri.it/). Data were extracted, anonymized, and compiled in Excel. Consent for scientific data use is part of the standard PDT consent process. The study followed the International Conference on Harmonisation Good Clinical Practice guidelines and the 2008 Declaration of Helsinki provisions. This manuscript was prepared in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology guidelines for observational studies. In our ICU, clinical decisions for PDT in oro-tracheal intubated patients were based on two criteria: 1) difficulty weaning from mechanical ventilation (three failures of the spontaneous breathing trial); 2) prediction of prolonged LOS, as seen in traumatic brain injury or cerebrovascular accidents.
Two experienced intensivists performed PDT under sterile conditions using a Blue Rhino tracheostomy set with a fiberoptic bronchoscope. In our ICU, all mechanically ventilated patients receive continuous sedation from the time of orotracheal intubation, according to standardized protocols, typically with propofol or midazolam in combination with short-acting opioids such as remifentanil, titrated to Richmond Agitation-Sedation Scale (RASS) targets and ventilator synchrony. Prior to initiating the PDT procedure, sedation was adjusted as needed to ensure RASS-5, and all patients additionally received rocuronium according to the unit protocol.
Data collected at ICU admission included demographics, comorbidities, source and reason for ICU stay, pre-ICU procedures, sepsis or trauma at entry, ICU LOS, and survival. To stratify patients’ clinical conditions, we calculated the Charlson Comorbidity Index (CCI), Sequential Organ Failure Assessment (SOFA) Score, and Glasgow Coma Scale (GCS). We reported the day from ICU admission to PDT and the LOS from PDT to ICU discharge. PDT timing was categorized as early (≤ 7 days), intermediate (8–14), or late (> 14).
Venous blood was sampled daily at 6–8 am. Laboratory values were collected at baseline (prePDT) and 24, 48, 72, and 96 hours after. In cases of multiple daily values, the mean was used. Using laboratory data, we computed the following indices: NLR [10], PLR [13], monocyte-to-lymphocyte ratio (MLR) [9], SIRI [11], Systemic Immune-Inflammation Index (SII) [12], and Aggregate Index of Systemic Inflammation (AISI) [14]. Supplementary File 1 (Section 1) provides details for all variables collected.
Exclusion criteria were: age < 18 years old, COVID-19 and H1N1 influenza, time from in-hospital to ICU admission > 0 days, PDT performed within 2 days from ICU admission, and post-PDT LOS in ICU ≤ 3 days. Then, we evaluated missing data (MD). MD were considered as missing completely at random, and observations were deleted.
The local ethics committee (Campania 2) approved the study (ID 2025/8030), waiving informed consent.
Statistical analyses
Statistical analyses were performed using R-Studio (version 2024, Posit Software). Categorical variables were reported as absolute numbers and percentages (%). Continuous data were assessed for normal distribution using the Shapiro-Wilk test. Normally distributed continuous data were expressed as mean ± standard deviation, while non-normally distributed data were expressed as the median and first-third quartile (Q1–Q3). Laboratory values and index variables at Time0 were divided into tertiles, and the groups were labeled as “Low,” “Medium,” and “High.” To compare survivors and non-survivors, we used the χ2 test for categorical variables, and Student’s t-test (or Welch’s t-test in the case of heteroscedasticity) for normally distributed continuous data. Non-normally distributed data were analyzed with the Mann-Whitney U test. Time trends were assessed using analysis of variance (ANOVA) for repeated measures or the Friedman test. When significant, pairwise comparisons were performed using paired t-tests or Wilcoxon signed-rank tests, with false discovery rate correction. Time0 served as the reference point.
To analyze the difference between tertiles over time, we performed two-way ANOVA for repeated measures or, in the case of skewed data, the Aligned Rank Transform (ART) method. Post-hoc analyses were conducted according to the results (see Supplementary File 1, Section 2). Mortality differences among tertiles were assessed using the χ2 test. We then performed univariate and multivariate logistic regression, adjusting for CCI and SOFA due to their prognostic relevance [15]. Results were reported as odds ratios (OR) with 95% confidence intervals (95% CI). For significant associations, Kaplan-Meier (KM) curves were generated using post-PDT days, and survival differences were tested with the log-rank test.
Results were reported in tables and plots. All tests were performed with a = 0.05, and a P-value < 0.05 was considered significant.
RESULTS
From 1st January 2020 to 10th June 2024, 1211 patients were admitted to the ICU, of whom 176 (14.5%) underwent PDT. After applying exclusion criteria and deleting MD, we included 114 patients (Figure 1). Baseline characteristics are summarized in Table 1. Briefly, 73 patients (64.0%) were male, with a median age of 59.5 years (Q1–Q3 49.0–68.7) and a median body mass index (BMI) of 26.2 kg m–2 (Q1–Q3 24.7–29.4). Thirty-nine patients (34.2%) were transferred from other hospitals, and 80 patients (70.2%) were previously admitted to the emergency department. The median ICU LOS was 42.5 days (Q1–Q3 27.20–60.0), with an ICU survival rate of 76.3% (87 patients). The most common comorbi dities were hypertension (82 patients, 71.9%), obesity (25 patients, 21.9%), and COPD (14 patients, 12.3%). The median CCI was 2.0 points (Q1–Q3 1.0–3.7), and the median SOFA and GCS scores at ICU admission were 10.0 points (Q1–Q3 7.0–12.0) and 5.0 (Q1–Q3 4.0–6.0), respectively. Trauma was the reason for ICU admission in 29.8% of cases (34 patients), while infection was present at admission in 4.4% (five patients). Neurosurgical procedures had been performed prior to ICU admission in 47.4% of patients (55 patients), and 24.6% underwent neuroradiological interventions (28 patients). All PDTs were complication-free. The median time from ICU admission to PDT was 14.0 days (Q1–Q3 10.2–19.0). Early, intermediate, and late PDT were performed, respectively, in 15 (13.1%), 44 (38.6%), and 55 patients (48.2%). The median post-PDT LOS was 27.0 days (Q1–Q3 14.0–44.0).
TABLE 1
Main population characteristics (n = 114)
[i] Categorical variables are reported as absolute number and percentage (%). Continuous variables are reported as median, first and third quartile (Q1–Q3) distribution, minimal (Min) and maximal (Max) values.
[ii] AISI – Aggregate Index of Systemic Inflammation, BMI – body mass index, CCI – Charlson Comorbidity Index, GCS – Glasgow Coma Scale, ICU – intensive care unit, LOS – length of stay, MLR – monocyte-to-lymphocyte ratio, NLR – neutrophil-to-lymphocyte ratio, PDT – percutaneous dilatational tracheostomy, PLR – platelet-to-lymphocyte ratio, SII – Systemic ImmuneInflammation Index, SIRI – Systemic Inflammation Response Index, SOFA – Sequential Organ Failure Assessment, WBC – white blood cells
Table 2 compares survivors and non-survivors. Non-survivors were older (63.0 vs. 59.0 years) and had a higher BMI (27.5 vs. 26.1 kg m–2) and a greater comorbidity burden (CCI 3.0 vs. 2.0; all P < 0.001). Trauma was more frequent in survivors (35.6% vs. 11.1%, P = 0.028), while infection at admission was higher in non-survivors (14.8% vs. 1.1%, P = 0.013). SOFA and GCS medians were similar, but distributions differed significantly (both P < 0.001). Timing of PDT differed slightly between groups, with a longer delay observed in non-survivors (median 15.0 vs. 14.0 days, P < 0.001) and a shorter post-PDT ICU stay (22.0 vs. 28.0 days, P < 0.001). At baseline (Time0), non-survivors had significantly lower values of total white blood cells (WBC), neutrophils, lymphocytes, monocytes, hemoglobin (Hb), and platelets (all P < 0.001). Regarding derived indices, the non-survivor group showed higher NLR (median 8.8 vs. 7.9, P < 0.001) but lower PLR, MLR, SIRI, SII, and AISI (all P < 0.001). Significant temporal variations were observed for several variables (see Supplementary File 1, Section 3.1). Specifically, WBC (P = 0.002), neutrophils (P = 0.005), platelets (P = 0.004), NLR (P = 0.046), MLR (P = 0.027), SIRI (P = 0.013), and AISI (P = 0.039) showed statistically significant changes over time. No significant trends were found for lymphocytes, Hb, PLR, or SII.
TABLE 2
Statistical comparisons of survivors vs. non-survivors
Pairwise comparisons revealed that WBC increased from Time0 (median 10.7 × 103 units mL–1) to Post48 (median 11.1 × 103 units mL–1, P = 0.044), and platelets increased from Time0 (median 280.5 × 103 units mL–1) to Post24 (median 289.5 × 103 units mL–1, P = 0.041), Post72 (median 294.0 × 103 units mL–1, P = 0.032), and Post96 (median 307.4 × 103 units mL–1, P = 0.036). AISI (Time0 median 1626.6) rose significantly at Post48 (median 1694.7, P = 0.024). Pairwise differences for NLR, MLR, and SIRI lost significance after correction.
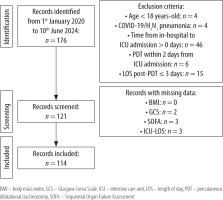
Supplementary File 1 (Section 3.2) provides a detailed description of the results. Only monocytes and AISI showed significant tertile–time interactions associated with outcomes (Table 3). For monocytes (Figure 2A), post-hoc analysis with the Friedman test showed that only trends in the Low tertile (χ2 = 32.600, df = 4, P < 0.001) were statistically significant. The pairwise analysis reported a statistically significant difference from Time0 for all consecutive times (paired W test P < 0.001), suggesting a constant increase over time. Mortality rates were higher in the Low group (36.8%, 14 patients), but not statistically significantly (χ2 = 5.534, P = 0.063). In the univariate logistic regression analysis, patients in the High tertile showed a reduced death probability compared to Low tertiles (OR: 0.321, 95% CI: 0.302-0.925, P = 0.042). In the multivariate model, Medium (OR: 0.323, 95% CI: 0.101–0.937, P = 0.044) and High (OR: 0.287, 95% CI: 0.087–0.847, P = 0.029) tertiles showed a reduced death probability compared to the Low tertile. The 30-day survival probabilities were 71.0% (95% CI: 55.6–90.6%) for Low, 87.1% (95% CI: 76.0–99.9%) for Medium, and 92.1% (95% CI: 83.9–100.0%) for High. The median survival times could not be determined for any group, as less than 50% of patients experienced the event within the study period. The log-rank test indicated no significant difference in survival curves among tertiles (χ2 = 5.600, df = 2, P = 0.061, Figure 3A). For AISI (Figure 2B), post-hoc analysis with the Friedman test showed that trends in Low (χ2 = 22.600, df = 4, P < 0.001), Medium (χ2 = 11.900, df = 4, P = 0.018), and High (c 2 = 20.700, df = 4, P < 0.001) tertiles were statistically significant. The Low tertile had a consistent increase, with significant pairwise changes (paired W test P < 0.05). In the Medium tertile, only the difference between Time0 and Post48 was significant (paired W test P = 0.006). In the High tertile, a significant difference was observed only between Time0 and Post72 (paired W test P = 0.042). Death rates in tertiles were 31.6% (12 patients) in the Low, 26.3% (10 patients) in the Medium, and 13.1% (5 patients) in the High tertile. However, mortality rates were not statistically significant (χ2 = 3.785, P = 0.151). In the multivariate logistic regression analysis, High tertile (OR 0.270, 95% CI: 0.074–0.861, P = 0.034) was related to the outcome, but not in the univariate analysis (OR 0.328, 95% CI: 0.094–1.005, P = 0.060). The 30-day survival probabilities were 77.7% (95% CI: 64.3–93.7%) for the Low, 83.9% (95% CI: 71.7–98.2%) for the Medium, and 90.5% (95% CI: 80.5–100.0%) for the High tertile. The median survival times could not be determined for any group, as less than 50% of patients experienced the event within the study period. The log-rank test indicated no significant difference in survival curves among tertiles (χ2 = 3.000, df = 2, P = 0.200, Figure 3B).
TABLE 3
Tertile trends analysis. The table shows results of the tertile–time interaction analyzed with the aligned rank transform (ART) method
FIGURE 2
Tertile trend lines with median values for monocytes (A) and Aggregate Index of Systemic Inflammation (AISI) (B) over time. Dots represent median values, while shaded areas indicate the 95% confidence interval (95% CI)
FIGURE 3
Kaplan-Meier survival curves stratified by tertiles, with risk tables, for monocytes (A) and Aggregate Index of Systemic Inflammation (AISI) (B). Log-rank test showed that differences in survival curves for monocytes (χ2 = 5.600, df = 2, P = 0.061) and AISI (χ2 = 3.000, df = 2, P = 0.200) were not statistically significant

DISCUSSION
In this retrospective exploratory study, we evaluated the temporal behavior and prognostic relevance of laboratory inflammatory markers and derived indices in ICU patients undergoing PDT. Among several parameters, monocyte count and AISI were most strongly associated with ICU mortality, especially in the lowest tertiles. Both parameters also showed consistent post-procedural increases, particularly in the Low tertile groups. However, no significant differences in 30-day survival curves were observed among tertiles based on KM survival analysis. Other indices, although statistically significant in trend analyses, did not show a robust association with mortality.
Although PDT is less invasive than surgical tracheostomy, it can still trigger an inflammatory reaction characterized by temporal changes in laboratory and inflammatory indices. The clinical implications of these findings are manifold. First, the observed monocyte and AISI trends underscore their potential as dynamic markers for monitoring inflammatory responses after PDT. Patients classified within the Low tertile for these indices may represent a higher-risk subgroup, warranting closer surveillance and potentially more tailored therapeutic interventions. For instance, patients with low monocyte counts or AISI values might benefit from targeted immunomodulatory therapies or intensified supportive measures. Additionally, the practicality of these indices lies in their derivation from routine blood tests, offering a cost-effective and easily accessible method for risk stratification in the ICU. This aspect is particularly valuable in resource-limited settings, where advanced diagnostic tools may not be readily available.
Acute critical illness induces significant changes in the immune system, characterized by a complex interplay between hyperactivation and suppression of immune responses. This dysregulation can lead to immediate and long-term consequences, including organ dysfunction and increased infection susceptibility. The immune alterations in critically ill patients are multifaceted, involving innate and adaptive immune components, and are influenced by factors such as the underlying cause of illness, patient demographics, and comorbidities. Acute critical illness often triggers an exaggerated innate immune response, characterized by the release of pro-inflammatory cytokines, known as a cytokine storm, which can lead to tissue damage and organ failure [16, 17]. Conversely, critically ill patients often exhibit lymphopenia and a reduced capacity to produce cytokines [18, 19], with increased expression of inhibitory molecules [20].
Our findings suggested that monocyte count, like the AISI score, could be interpreted as a clinical manifestation of the immune system’s health status. Patients with a low monocyte count or AISI may likely represent particular patient phenotypes characterized either by a particular cytokine profile or by the occult presence of infectious conditions. The execution of PDT, triggering an inflammatory response, can then highlight particularly reactive profiles that actually underlie a dysfunction of the immune system. In fact, monocytes and AISI Low tertiles showed a significant and constant increase in values over time, not observed in High and Medium tertiles.
The balance between pro-inflammatory and anti-inflammatory responses is critical, and interventions must often be tailored to the individual patient’s immune status. Our study showed no difference in PDT timing frequency between surviving patients and those who died. Until now, the literature has consistently questioned whether the timing of PDT could influence clinical outcomes such as mortality, ICU LOS, duration of sedation, and VAP incidence. However, the most recent meta-analyses have reported conflicting conclusions. Merola et al. [5] found that early tracheostomy slightly reduced mortality, ICU LOS, and duration of mechanical ventilation compared with late tracheostomy. However, early tracheostomy did not reduce the duration of mechanical ventilation compared with prolonged intubation and did not affect VAP. Furthermore, trial sequence analysis for each outcome did not find conclusive evidence, suggesting that further studies were needed. Han et al. [6] reported that early tracheostomy reduces the duration of mechanical ventilation and ICU LOS but did not significantly reduce the incidence of VAP and mortality. Premraj et al. [7], in a meta-analysis of over 17000 critically ill stroke patients, demonstrated that the timing of tracheostomy was not associated with mortality, neurological outcomes, or ICU/hospital LOS.
Monocytes are integral to innate and adaptive immune systems and play a central role in the immune response of critically ill patients, particularly in the context of sepsis and septic shock [21]. Their dysfunction, including reduced expression of human leukocyte antigen–DR isotype, contributes to immunoparalysis and increased infection risk [22, 23]. While monocytes are typically categorized into classical, intermediate, and non-classical subsets [24], the association between monocyte trends and outcomes in patients undergoing PDT remains unexplored.
Hortová-Kohoutková et al. [25] demonstrated that specific monocyte subsets correlate with cytokine levels and survival in septic shock. Based on our findings, it is conceivable that survival differences across tertiles may be influenced by the distribution of monocyte subtypes rather than absolute counts. Future studies should investigate this hypothesis, focusing on subset dynamics over time.
The AISI, derived from neutrophil, monocyte, platelet, and lymphocyte counts, is gaining attention as a prognostic marker in various clinical settings. Its utility in critical care has been supported by studies in COVID-19, where higher AISI values were associated with disease severity and mortality, with significant sensitivity and specificity for these outcomes [26]. Tarle et al. [27], in a retrospective study, compared AISI with other systemic inflammatory indices such as the C-reactive protein (CRP) and the SII index. In the context of odontogenic abscesses, AISI was found to be a superior predictor of abscess severity compared to CRP, demonstrating higher sensitivity and specificity. Although AISI is often associated with poor outcomes, our data suggest that low values may reflect impaired immune competence and limited inflammatory reserve. To our knowledge, this inverse association has not been previously reported and warrants further investigation.
While the strengths of this study include its focus on a standardized procedural context and the use of validated systemic inflammation indices, several limitations must be acknowledged.
First, its retrospective design limits causal inference, and the small sample size may have reduced the power to detect survival differences. Confounders such as nutritional status or undiag-nosed infections were not captured. Furthermore, detailed information on vasopressor and antibiotic exposure was not collected. These therapies are frequently administered in critically ill patients and may influence bone marrow function, leukocyte kinetics, and systemic inflammatory indices [28, 29]. Second, the analysis was restricted to the first 96 hours after PDT, preventing assessment of long-term inflammatory trends or complications such as late-onset sepsis. Third, being a single-center study, external validity is limited. Finally, only ICU mortality and short-term outcomes were analyzed; longerterm endpoints such as 90-day survival were not assessed.
CONCLUSIONS
This exploratory study highlights the potential role of monocytes and AISI as accessible tools to monitor inflammation and stratify risk in ICU patients undergoing PDT. These indices may support clinical decision-making but should be interpreted alongside a comprehensive evaluation. Rather than focusing on PDT timing, future research should assess patients’ immunological profiles. Larger, prospective studies with immune phenotyping are needed to validate these findings and clarify the impact of low inflammatory reserve on outcomes.